X-RAY DIFFRACTION (XRD) FOR BATTERY
DETERMINE THE THREE DIMENSIONAL STRUCTURE OF MATTER
X-ray diffraction (XRD) is one of the most important non-destructive tools for the analysis of all kinds of matter—ranging from fluids to powders and crystals. Widely utilized in all aspects of materials development, starting with early research through to final production and engineering, XRD is an indispensable method for materials structural characterization and quality control. Rigaku has developed a range of X-ray diffractometers, in cooperation with academic and industrial users, which provide the most technically advanced, versatile, and cost-effective diffraction solutions available today.
For materials containing crystalline components, XRD is routinely utilized for crystalline phase identification and subsequent quantitation. Identification relies on the comparison of measured diffraction patterns against databases of known materials. For novel materials, databases can be built on the fly to monitor any material changes during development. Quantitative analysis can be based on known reference materials or novel materials with known elemental composition.
In addition to fingerprinting, identification, and quantitation, X-ray diffraction techniques are the gold standard in elucidating the three-dimensional atomic/molecular structure of crystalline and non-crystalline materials. As the properties of a material are essentially governed by its inherent atomic-level structure, X-ray diffraction techniques are widely utilized and indispensable for materials research, development, and production.
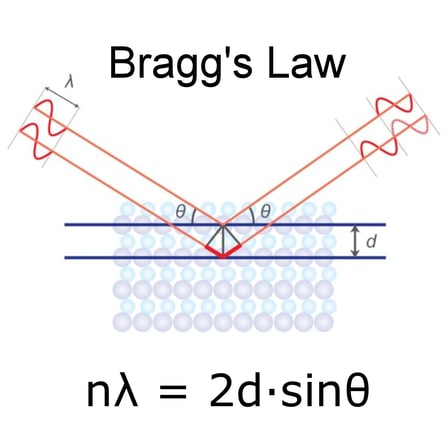
For crystalline solids, the Bragg equation nλ = 2dsinθ is one of the keystones in understanding X-ray diffraction. In this equation, n is an integer, λ is the characteristic wavelength of the X-rays impinging on the crystallized sample, d is the interplanar spacing between rows of atoms, and θ is the angle of the X-ray beam with respect to these planes. When this equation is satisfied, X-rays scattered by the atoms in the plane of a periodic structure are in phase and diffraction occurs in the direction defined by the angle θ. In the simplest instance, an X-ray diffraction experiment consists of a set of diffracted intensities and the angles at which they are observed. This diffraction pattern can be thought of as a structural fingerprint, and structural identification can be performed by comparing this diffraction pattern to a database of known patterns.
Although less well known, the Debye diffraction equation plays a similarly pivotal role for non-crystalline materials as the Bragg equation plays for crystalline materials. The Debye approach to diffraction led to the development of Pair Distribution Function (PDF) analysis and Total Diffraction which have become indispensable for the analysis of inherent local structure in disordered systems.
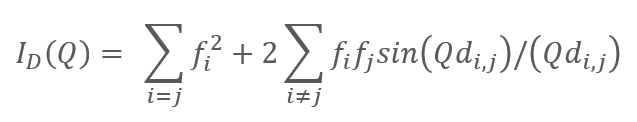
The Debye diffraction equation where Q is the diffraction vector, di,j is the distance between atom pairs and fi is the individual atomic form factor (number of electrons).


.jpg)

.jpg)





-1.jpg?width=700&height=1000&name=B-XRD1123_Simultaneous_Operando_XRD_measurement_for_positive_and_negative_electrode_---_ApplicationNote_Q1121en_700x1000%20(1)-1.jpg)

.jpg?width=700&height=1000&name=B-XRD1124_Operando_transmission_XRD_measurement_of_all-solid-state_litium-ion_battery_using_Ag_source_ApplicationNote_R0727en_700x1000%20(1).jpg)

.jpg?width=700&height=1000&name=B-XRD1116_Operando_measurement_of_laminated_litium_ion_battery_using_2DD_ApplicationNote_P0306en_700x1000%20(1).jpg)




